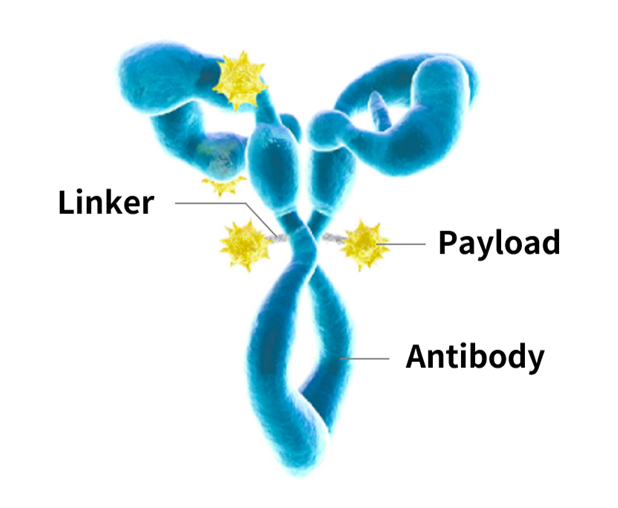
Antibody-drug conjugates (ADCs) symbolize the latest approach in cancer therapy, merging the aiming ability of monoclonal antibodies with the strength of cytotoxic doses. This advanced treatment mode is directed at destructing cancer cells, although it spars sound tissues selectively. The success of ADCs heavily depends on ADC Bioanalysis, a particularized field concentrated on assessing the pharmacokinetics (PK), pharmacodynamics (PD), immunogenicity, and constancy of these complicated molecules. In this article, we will review ADC Bioanalysis’s key aspects and analytic techniques. Let’s explore more.
Contents
Key Aspects of ADC Bioanalysis:
Here are some key aspects of ADC Bioanalysis.
1. Pharmacokinetics (PK) Analysis:
Pharmacokinetics involves analysing the movement of doses within the body. Due to their double nature—representing both an antibody and a cytotoxic payload—ADCs’ PK analysis is especially complicated. Advanced bioanalytical methods are employed to assess the absorptions of the entire ADC, the free antibody, and the free drug in different natural matrices.
2. Pharmacodynamics (PD) Studies:
Pharmacodynamics concentrates on the drug’s consequences on the human body. Bioanalytical checks are developed to evaluate the fundamental interaction of ADCs with objective cells, their adhering affinity, and the leading cellular reactions. These analyses help foretell the healthful efficacy and expected fallouts of ADCs.
3. Immunogenicity Assessment:
Immunogenicity cites the potency of ADCs to set off an immune reaction in the body, which can affect their safety and efficacy. Bioanalytical methods are utilized to discover and measure anti-drug antibodies (ADAs) that might develop in ADC administration reactions. Supervising immunogenicity is critical for controlling patient safety and dealing with adverse responses.
4. Consistency Testing:
Consistency testing estimates the respectability and strength of ADCs in more favourable conditions. Given their confounded structure, ADCs can debase over the long run, prompting diminished viability or expanded toxicity. Bioanalytical techniques are utilized to assess the steadiness of ADCs during capacity, taking care of, and administrations.
Analytical Techniques in ADC Bioanalysis:
A lot of advanced analytical techniques are utilized in ADC Bioanalysis to accomplish exact measurements:
· Liquid Chromatography-Mass Spectrometry (LC-MS):
LC-MS is a strong strategy for estimating and qualifying ADCs and their components. It offers high awareness and explicitness, guaranteeing the recognition of low-portion retention in convoluted biologic matrices.
· Enzyme-Linked Immunosorbent Assay (ELISA):
ELISA is broadly used for measuring antibodies and ADAs. A very touchy and specific strategy can distinguish and evaluate the presence of ADCs and their immunogenic responses in organic examples.
· Capillary Electrophoresis (CE):
CE is utilized for the interval and characterization of ADCs based on size and charge. It is beneficial for evaluating the heterogeneity and purity of ADC preps.
Importance of ADC Bioanalysis in Drug Development:
ADC Bioanalysis is a base of ADC drug development. It allows vital data that leads decision-making throughout the development procedure, from presymptomatic analyses to clinical tests and regulatory compliances. The insights acquired from bioanalytical analyses assist in optimizing ADC design, bettering therapeutic efficacy, assuring patient safety, and coping with regulatory demands.
Last Words:
ADC Bioanalysis is a fundamental field that upholds the development of Antibody-Drug Conjugates. Through novel logical strategies and demanding approaches, researchers can guarantee that ADCs are secure, successful, and legitimate therapies for malignant growth. As ADC innovative work keep on enhancing, ADC Bioanalysis will persevere as an exceedingly significant factor getting these high-level treatments to patients in pain.